| BSA conjugation on N terminal -NH2 | KLH conjugation on N terminal -NH2 | OVA conjugation on N terminal -NH2 | Acetylation |
| Biotin | Biotin-Ahx | 5-FAM | 5-FAM-Ahx |
| 6-FAM | 6-FAM-Ahx | 2-Abz | 4-Abz |
| Cy3 | Cy5 | Cy5.5 | Cy7 |
| DABCYL | Dansyl | Dansyl-Ahx | FITC-Ahx |
| FITC-PEG2 | 5-TMR | 6-TMR | Rhodamine B |
| MCA | 3-Maleimide | 6-Maleimide | SMCC |
| Acryl | Alloc | Benzoyl | CBZ |
| Fmoc | Br-Ac | Cl-Ac | Aminooxy |
| DOTA | NOTA | 1-Nap | 2-Nap |
| Succinylation | Glutaric acid | Butyric acid,C4 | Hexanoic acid,C6 |
| Octanoic acid,C8 | Nonanoic acid,C9 | Decanoic acid,C10 | Lauric acid,C12 |
| Myristic acid,C14 | Palmitic acid,C16 | Stearic acid,C18 | Arachidic acid,C20 |
| L-Lactic acid | D-Lactic acid | (R)-Lipoic acid | 2-Mercaptoacetic acid |
| 2-Azidoacetic acid | 4-Azidobutyric acid | 6-Azidohexanoic acid | Propiolic acid |
| 5-Hexynoicacid | Trans-Cinnamic acid | Trans-Crotonic acid | DBCO |
| Methyltetrazine | TCO |
-
REAGENT SERVICES
Hot!
-
Most Popular Services
-
Molecular Biology
-
Recombinant Antibody/Protein
-
Reagent Antibody
-
CRISPR Gene Editing
-
DNA Mutant Library
-
IVT RNA and LNP Formulations
-
Oligo Synthesis
-
Peptides
-
Cell Engineering
-
- Gene Synthesis FLASH Gene
- GenBrick™ Up to 200kb
- Gene Fragments Up to 3kb now
- Plasmid DNA Preparation Upgraded
- Cloning and Subcloning
- ORF cDNA Clones
- mRNA Plasmid Solutions New!
- Cell free mRNA Template New!
- AAV Plasmid Solutions New!
- Mutagenesis
- GenCircle™ Double-Stranded DNA New!
- GenSmart™ Online Tools
-
-
PRODUCTS
-
Most Popular Reagents
-
 Instruments
Instruments
-
Antibodies
-
ELISA Kits
-
Protein Electrophoresis and Blotting
-
Protein and Antibody Purification
-
Recombinant Proteins
-
Molecular Biology
-
Stable Cell Lines
-
Cell Isolation and Activation
-
 IVD Raw Materials
IVD Raw Materials
-
 Therapy Applications
Therapy Applications
-
Resources
-
- All Instruments
- Automated Protein and Antibody Purification SystemNew!
- Automated Plasmid MaxiprepHot!
- Automated Plasmid/Protein/Antibody Mini-scale Purification
- eBlot™ Protein Transfer System
- eStain™ Protein Staining System
- eZwest™ Lite Automated Western Blotting Device
- CytoSinct™ 1000 Cell Isolation Instrument
-
- Pharmacokinetics and Immunogenicity ELISA Kits
- Viral Titration QC ELISA Kits
- -- Lentivirus Titer p24 ELISA KitHot!
- -- MuLV Titer p30 ELISA KitNew!
- -- AAV2 and AAVX Titer Capsid ELISA Kits
- Residual Detection ELISA Kits
- -- T7 RNA Polymerase ELISA KitNew!
- -- BSA ELISA Kit, 2G
- -- Cas9 ELISA KitNew!
- -- Protein A ELISA KitNew!
- -- His tagged protein detection & purification
- -- dsRNA ELISA Kit
- -- Endonuclease ELISA Kit
- COVID-19 Detection cPass™ Technology Kits
-
- Automated Maxi-Plasmid PurificationHot!
- Automated Mini-Plasmid PurificationNew!
- PCR Reagents
- S.marcescens Nuclease Benz-Neburase™
- DNA Assembly GenBuilder™
- Cas9 / Cas12a / Cas13a Nucleases
- Base and Prime Editing Nucleases
- GMP Cas9 Nucleases
- CRISPR sgRNA Synthesis
- HDR Knock-in Template
- CRISPR Gene Editing Kits and Antibodies
-
![AmMag™ Quatro Automated Plasmid Purification]() AmMag™ Quatro automated plasmid purification
AmMag™ Quatro automated plasmid purification
-
![Anti-Camelid VHH]() MonoRab™ Anti-VHH Antibodies
MonoRab™ Anti-VHH Antibodies
-
![ELISA Kits]() ELISA Kits
ELISA Kits
-
![Precast Gels]() SurePAGE™ Precast Gels
SurePAGE™ Precast Gels
-
![Quatro ProAb Automated Protein and Antibody Purification System]() AmMag™ Quatro ProAb Automated Protein and Antibody Purification System
AmMag™ Quatro ProAb Automated Protein and Antibody Purification System
-
![Target Proteins]() Target Proteins
Target Proteins
-
![AmMag™ Quatro Automated Plasmid Purification]() AmMag™ Quatro automated plasmid purification
AmMag™ Quatro automated plasmid purification
-
![Stable Cell Lines]() Stable Cell Lines
Stable Cell Lines
-
![Cell Isolation and Activation]() Cell Isolation and Activation
Cell Isolation and Activation
-
 IVD Raw Materials
IVD Raw Materials
-
![Quick
Order]() Quick Order
Quick Order
-
![Quick
Order]() Quick Order
Quick Order
- APPLICATIONS
- RESOURCES
- ABOUT US
- SIGN IN My Account SIGN OUT
- REGISTER
Hi!Ask me about GenScript services and products! I can answer questions or connect you to a live person.
![Peptide Modifications Peptide Modifications]()
Peptide Modifications
Over 300 modifications; Free amidation and acetylation Includes
biotin, FITC, PEGylation, methylation, disulfide bonds KLH, BSA, OVA conjugationsHome » Peptide Services » Peptide Modifications
Overview
The peptide modification services at GenScript offer a wide range of modifications to meet any research need. These modifications can improve overall peptide stability, alter structure to better understand biological function, or enhance immunogenicity for antibody development and production. In addition to a variety of terminus and internal modifications, GenScript's services include peptide labeling and conjugations for imaging and detection needs.
For a list of our available modifications, select the modification options below
-
N-terminal modifications
-
C-terminal modifications
Amidation Alcohol AMC Bzl Cysteamide EDA-Biotin Ester (OEt) Ester (OMe) Glu(EDANS) Hydrazine MPAA NHEt Nhisopen NHMe TBzl p-Nitroanilide Tyr (3-NO2) BSA conjugation on C terminal -COOH KLH conjugation on C terminal -COOH OVA conjugation on C terminal -COOH -
Special amino acids
-
Stable isotope labeled peptides
{Arg(13C6,15N4)} {Ile(13C6,15N)} {Leu(13C6,15N)} {Lys(13C6,15N2)} {Phe(13C9,15N)} {Pro(13C5,15N)} {Val(13C5,15N)} -
Fluorescent peptide modifications/FRET pairs
2-Abz (N-Terminal) FITC-Ahx (N-Terminal) 4-Abz (N-Terminal) FITC-PEG2 (N-Terminal) 5-FAM (N-Terminal) MCA (N-Terminal) 5-FAM-Ahx (N-Terminal) Rhodamine B (N-Terminal) 6-FAM (N-Terminal) AMC (C-Terminal) 6-FAM-Ahx (N-Terminal) {ACC}
(7-Aminocoumarin-4-Acetic acid)5-TMR (N-Terminal) MCA/Lys(DNP) 6-TMR (N-Terminal) 2-Abz/Lys(DNP) Cy3 (N-Terminal) 4-Abz/Lys(DNP) Cy5 (N-Terminal) 2-Abz/Tyr(3-NO2) Cy5.5 (N-Terminal) 4-Abz/Tyr(3-NO2) Cy7 (N-Terminal) DABCYL/Glu(EDANS) Dansyl (N-Terminal) DABCYL/EDANS Dansyl-Ahx (N-Terminal) -
Peptide conjugates
BSA (-COOH of C terminal) BSA (-NH2 of N terminal) BSA Conjugation on cysteine KLH (-COOH of C terminal) KLH (-NH2 of N terminal) KLH Conjugation on cysteine OVA (-COOH of C terminal) OVA (-NH2 of N terminal) OVA Conjugation on cysteine -
Other modifications (MAPS, PEGylation, cyclic modifications)
MAPS PEGylation Cyclic modifications Disulfide Bridges Other MAPS Asymmetric 2 branches (C-Terminal) {PEG1}
NH2-(PEG)1-CH2COOHHead to tail amide cyclic Random Disulfide Bridge Dimer ( Inter-Disulfide bridge) MAPS Asymmetric 4 branches (C-Terminal) {PEG1-propionic acid}
NH2-PEG1-CH2CH2COOHAmide cyclic (Side chain) Mono Disulfide bridge MAPS Asymmetric 8 branches (C-Terminal) {PEG2}
NH2-(PEG)2-CH2COOHStapled peptide(S5/S5) Double Disulfide bridge {PEG3}
NH2-(PEG)3-CH2CH2COOHStapled peptide(R8/S5) Triple Disulfide Bridge {PEG4}
NH2-(PEG)4-CH2CH2COOHMono Thioether Bridge {PEG5}
NH2-(PEG)5-CH2CH2COOHThioester (C-terminal) {PEG6}
NH2-(PEG)6-CH2CH2COOHAmide cyclic (Head to side chain) {PEG8}
NH2-(PEG)8-CH2CH2COOHAmide cyclic (Side chain to tail) {PEG11}
NH2-(PEG)11-CH2COOH{PEG12}
NH2-(PEG)12-CH2CH2COOH -
Peptoid
Key Features (Peptoid VS. Peptide)
- More stable: Peptoids are less susceptible to degradation in vivo than peptides.
- More choices: Peptoids are well suited for combinatorial approaches to drug discovery because large libraries can be synthesized easily from readily available primary amines.
- More cost-effective and time-efficient: Major advances in screening methodology have allowed peptoid libraries of hundreds of thousands of compounds to be mined inexpensively and quickly for highly specific protein-binding.
- Higher market potential: The features of peptoids make them a class of pharmacological agents with great potential.
Service Specifications
Peptoid Synthesis(Cat. No.: SC1559)
- 20 different residues available, more in development
- Peptoid length up to 20 residues
- Peptoid purity from crude to ≥98%
- Peptoid quantity up to 500 mg
- Available modifications: Acetylation, Amidation, Biotin, FAM, FITC, TMR labeling
- MS, HPLC, and COA provided for each peptoid
- 2-6 weeks turnaround time, depending on the length and quantity of ordered peptoid
Current Available Peptoid Side Chain
Side Chain NameCodeStructural FormulaMethylamine{NAla}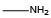 Ethylamine{NAbu}
Ethylamine{NAbu} n-propylamine{Nnpa}
n-propylamine{Nnpa} n-butylamine{Nnba}
n-butylamine{Nnba} Isopropylamine{NVal}
Isopropylamine{NVal}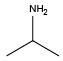 Sec-butylamine{NIle}
Sec-butylamine{NIle}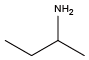 (S)-(+)-Sec-butylamine{(S)-NIle}
(S)-(+)-Sec-butylamine{(S)-NIle}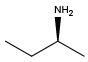 (R)-(-)-Sec-butylamine{(R)-NIle}
(R)-(-)-Sec-butylamine{(R)-NIle}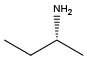 Isobutylamine{NLeu}
Isobutylamine{NLeu}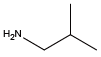 Benzylamine{NPhe}
Benzylamine{NPhe}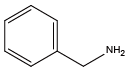 β-phenylethylamine{NPea}
β-phenylethylamine{NPea}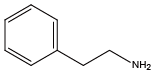 α-methylbenzylamine{Nmba}
α-methylbenzylamine{Nmba}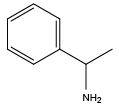 (S)-α-methylbenzylamine{(S)-Nmba}
(S)-α-methylbenzylamine{(S)-Nmba}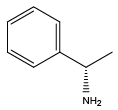 (R)-α-methylbenzylamine{(R)-Nmba}
(R)-α-methylbenzylamine{(R)-Nmba}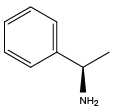 2-(methylthio)ethylamine{NMet}
2-(methylthio)ethylamine{NMet} 3-Methoxypropylamine{Nmpa}
3-Methoxypropylamine{Nmpa} 1,4-Butylenediamine{NLys}
1,4-Butylenediamine{NLys} 4-methoxyphenethylamine{Nmpe}
4-methoxyphenethylamine{Nmpe}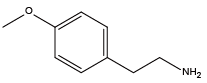 3,4-Dimethoxybenzylamine{Ndmb}
3,4-Dimethoxybenzylamine{Ndmb}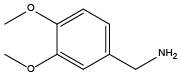 Tetrahydrofurfurylamine{Nffa}
Tetrahydrofurfurylamine{Nffa}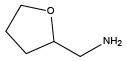 (S)-(+)-Tetrahydrofurfurylamine{(S)-Nffa}
(S)-(+)-Tetrahydrofurfurylamine{(S)-Nffa}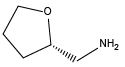 (R)-(-)-Tetrahydrofurfurylamine{(R)-Nffa}
(R)-(-)-Tetrahydrofurfurylamine{(R)-Nffa}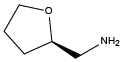 Piperonylamine{Npip}
Piperonylamine{Npip} 4-(2-Aminoethyl)benzenesulfonamide{Nbsa}
4-(2-Aminoethyl)benzenesulfonamide{Nbsa}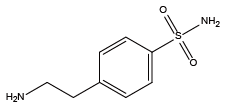 Tert-butylamine{Ntbu}
Tert-butylamine{Ntbu} Diisopropylamine{Ndip}
Diisopropylamine{Ndip}
N-terminal only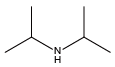 Tryptamine{NHtrp}
Tryptamine{NHtrp}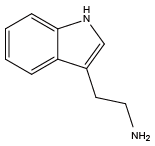 1H-Indole-3-methanamine{NTrp}
1H-Indole-3-methanamine{NTrp}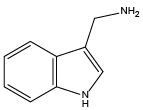 Cysteamine{NHcys}
Cysteamine{NHcys} 4-Hydroxybenzylamine{NTyr}
4-Hydroxybenzylamine{NTyr}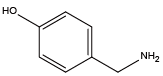 Histamine{NHhis}
Histamine{NHhis}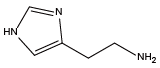 Glycine{NAsp}
Glycine{NAsp}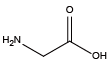 Glycinamide{NAsn}
Glycinamide{NAsn}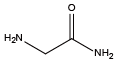 Beta-Alanine{NGlu}
Beta-Alanine{NGlu}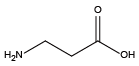 Beta-alanine amide{NGln}
Beta-alanine amide{NGln}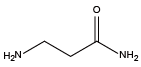
Delivery Specifications
The typical delivery package consists of lyophilized peptoids of required sequence, purity, quantity and associated QC reports. Every step of peptoid synthesis is subject to GenScript's stringent quality control.
* For your convenience, modifications can easily be added when you request a quote online. If you don't find the modification you are looking for,please contact [email protected] for support.
Peptide Modification Basics
Amidation and Acetylation
If the peptide is from an internal sequence of a protein, terminal amidation (C-terminus) or acetylation (N-terminus) will remove its charge and help it imitate its natural structure (amide, CONH2). In addition, this modification makes the resulting peptide more stable towards enzymatic degradation resulting from exopeptidases.
Biotin and FITC
For C-terminal labeling of biotin, a Lys residue is added to the C-terminus of the peptide. Biotin is then attached to the lysine side chain via amide bond. The positive charge of the lysine is then removed.
Fluorescein isothiocyanate (FITC) is an activated precursor used for fluorescein labeling. For efficient N-terminal labeling, a seven-atom aminohexanoyl spacer (NH2-CH2-CH2-CH2-CH2-CH2-COOH) is inserted between the fluorophore (fluoroscein) and the N-terminus of the peptide.
Disulfide Bridge
Peptide cyclization can be achieved through creating disulfide bridges between cysteine residues on the peptide. This is a challenging practice for peptide containing multiple cysteine residues due to random formations of disulfide bridges between them. GenScript is able to build disulfide bridges between cysteine at specified positions. We are able to introduce up to three customized disulfide bridges on one peptide.
Phosphorylation
Phosphopeptides can assist in the investigation of the influences of phosphorylation on peptides and protein structure and in the understanding of regulatory processes mediated by protein kinases. GenScript has successfully synthesized numerous serine-, threonine-, and tyrosine-phosphopeptides. For peptides containing one or more of these hydroxy-amino acids, selective phosphorylation can be achieved by orthogonal protection or by Fmoc-protected phosphorylated amino acids.
Methylation
The methylation of proteins has been established as an important modification that helps regulate cellular functions such as transcription, cell division, and cell differentiation. Post-translational N-methylation usually occurs on lysine or arginine sidechains. Peptides that represent methylated proteins are useful for protein-protein interaction studies or structural determination by x-ray crystallography. GenScript can synthesize peptides containing mono-, di-, and tri-methylated lysines at >98% purity, as well as other methylation combinations.
PEGylation
PEGylation is the covalent conjugation of macromolecules (antibody, peptide, etc.) with polyethylene glycol (PEG), polymers that are nonionic, nontoxic, biocompatible and highly hydrophilic. The PEGylated macromolecules have enhanced therapeutic properties due to their increased solubility (for hydrophobic drugs) and bioavailability, masked antigenicity for minimum immune response in host, prolonged circulatory time within host through reduced renal clearance.
Isotope Labeling
For NMR measurement, we can label peptides with stable nonradioactive isotopes. Peptides labeled with 2H, 15N, 13C, or both 15N and 13C can be synthesized for convenient detection in research.
MAPS
Multiple antigen peptide application is one potent way to produce high-titer anti-peptide antibodies and synthetic peptide vaccines. This system utilizes the α- and ε-amino groups of lysine to form a backbone to which multiple peptide chains can be attached. Depending on the number of lysine tiers, different numbers of peptide branches can be synthesized. This eliminates the need to conjugate the antigen to a protein carrier.
BSA, KLH, and OVA Conjugation
Peptide antigens are often too small to generate significant immune responses on their own. To solve this problem, these peptides are conjugated to larger carrier proteins, such as bovine serum albumin (BSA), ovalbumin (OVA), or keyhole limpet hemocyanin (KLH). KLH is advantageous because it does not interfere with ELISA or western blotting, as it is not used as a blocking reagent. A common conjugation method is the maleimide method, which couples the cysteine residue of the peptide to the carrier protein. To perform this conjugation, one cysteine residue is added to the N- or C-terminus of the peptide for linkage.
Note: KLH is a large aggregating protein (MW = 4×10⁵ to 1×10⁷) with limited water solubility, which gives solutions a cloudy appearance. However, this does not affect immunogenicity, and the turbid solution can still be used for immunizations. For shipments, peptides are sent as solutions chilled with blue ice if precipitates form after conjugation.
Related Services
Resources

Case Study
-
Fluorescent Labeled Peptides
-
Click Peptides
-
Peptoid
Fluorescent Modification Case studies
Fluorescent peptide labels have numerous research applications, and GenScript has extensive experience synthesizing peptides with a variety of modifications.
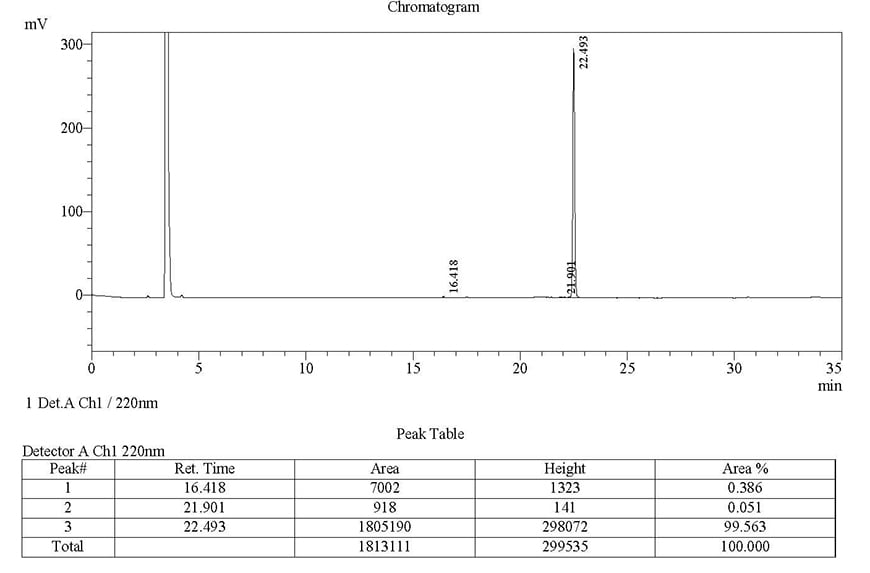
Case Study 1
Sequence: LYRLGLGH
Modification: MCA/DNP
Quantity: 1-4 mgRequired purity Estimated Turnaround time Actual purity Actual turnaround time >98%17 days99.563%14 daysClick here to view HPLC results »
Click here to view MS results »
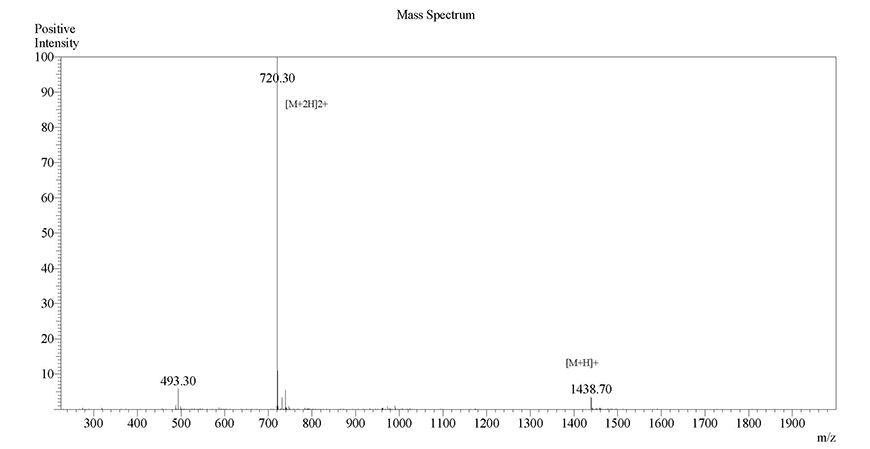
Case Study 2
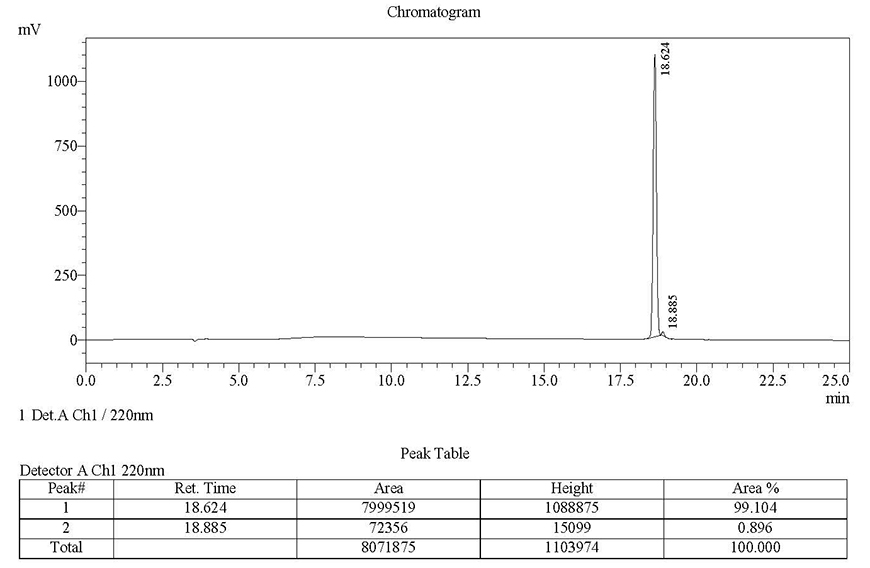
Sequence: IKDLSKEERLWEVQRILTALKRKLREA
Modification: 5-FAM (N-terminal)
Quantity: 10-14 mgRequired purity Estimated Turnaround time Actual purity Actual turnaround time >98%23 days99.10%13 daysClick here to view HPLC results »
Click here to view MS results »
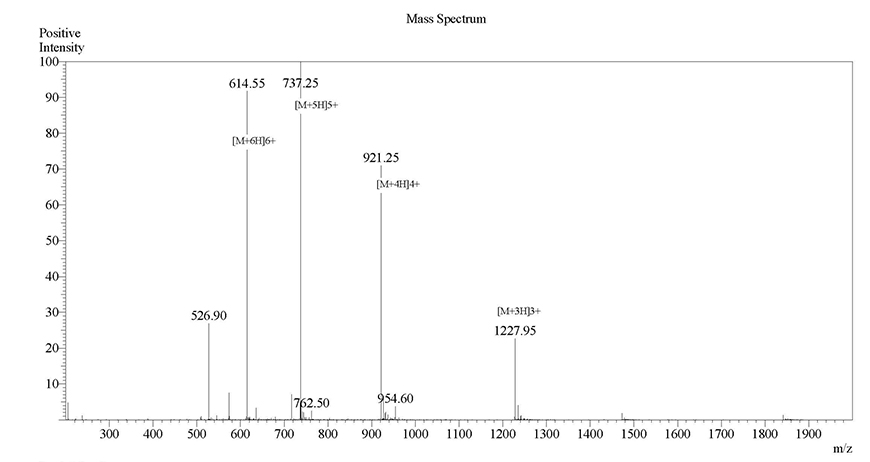
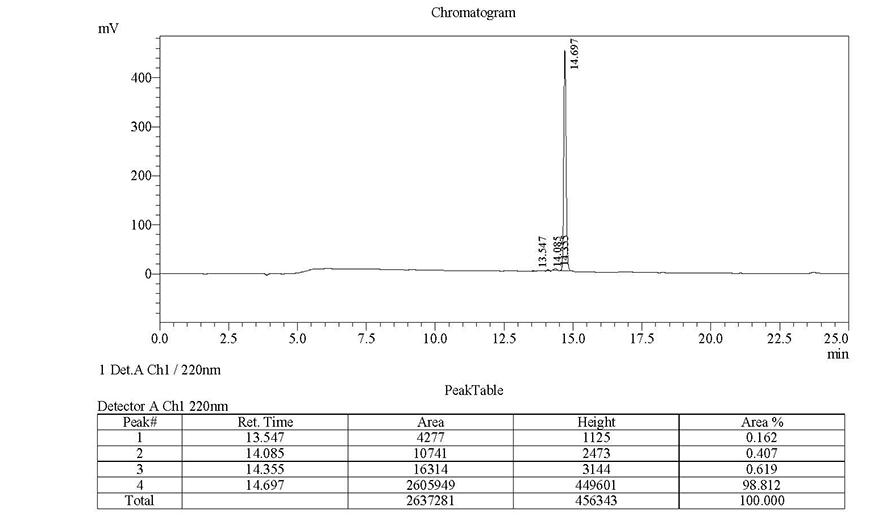
Case Study 3
Sequence: RAKWNNTLKQIASK
Modification: FITC-Ahx (N-terminal)
Quantity: 5-9 mgRequired purity Estimated Turnaround time Actual purity Actual turnaround time >98%17 days99.81%5 daysClick here to view HPLC results »
Click here to view MS results »
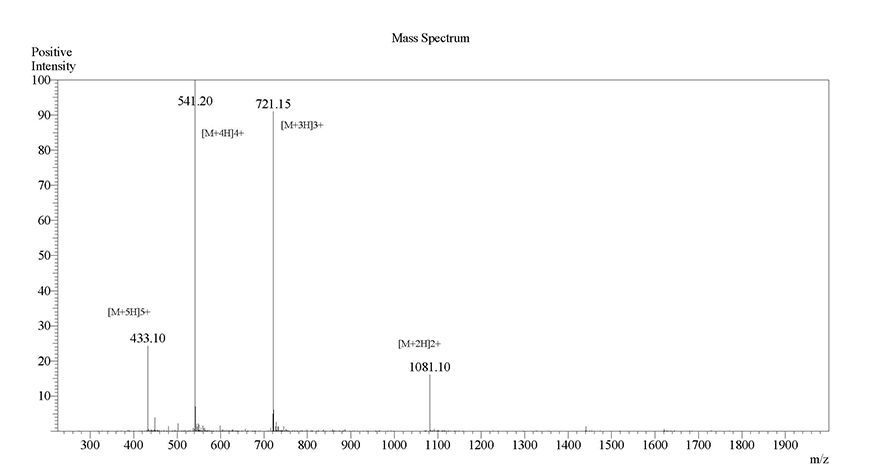
26-O-acyl β-amyloid (1-42) click peptide was successfully synthesized, the β-ester bond in which can be quickly and quantitatively converted to a native Gly25-Ser26 amide bond via a pH-dependent O-N intramolecular acyl migration reaction (t1/2 =1 min, pH7.4, 37℃) at a hydroxyamino acid residue. Namely, upon this pH-triggered conversion (pH-click), the non-aggregative and water-soluble precursor (click peptide) can produce the monomer with a random-coil structure under physiological conditions (pH7.4, 37℃). The structure information is as follows.
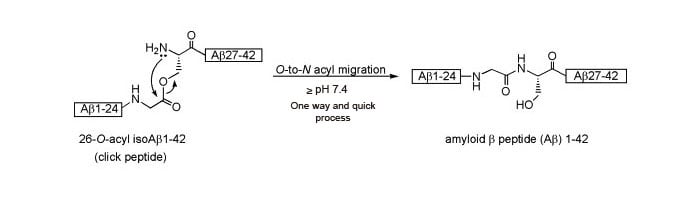
Scheme 2: β-amyloid (1-42) click peptide
Result
- β-amyloid precursor(click peptide) has a water solubility of 15 mg/ml, while it is only 0.14 mg/ml for the native peptide.
- The aggregative property of the peptides reduced significantly.
- The O-acyl moiety was stable under acidic pH.
The following are HPLC reports of (Scheme 3) purified β-amyloid (1-42) native peptide converted from β-amyloid (1-42) click peptide (precursor) and (Scheme 4) purified 26-O-acyl β-amyloid (1-42) click peptide (precursor).
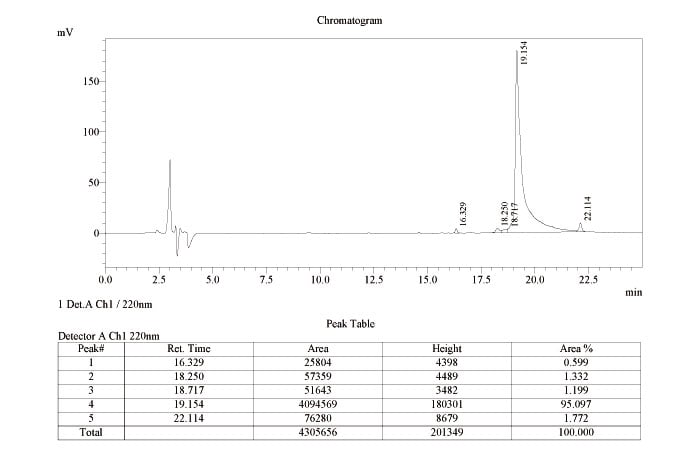
(Scheme 3) HPLC report of purified β-amyloid (1-42) native peptide converted from β-amyloid (1-42) click peptide (precursor)
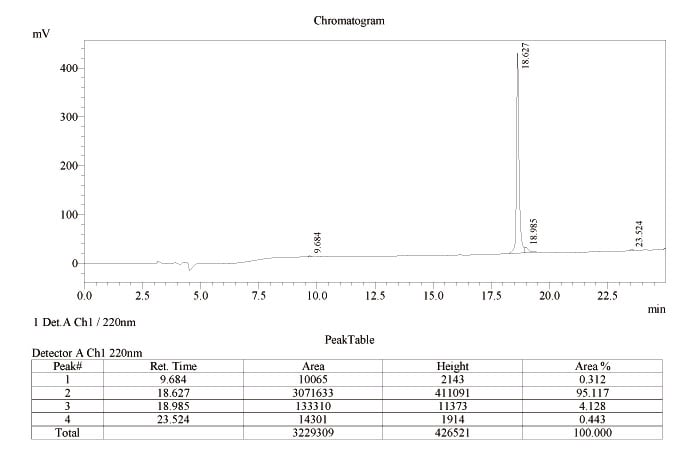
(Scheme 4) HPLC report of purified 26-O-acyl β-amyloid (1-42) click peptide (precursor)
Case Study
With proven technical capability and state-of-the-art in-house instruments and technologies, we have successfully synthesized many peptoids.
Modification: C-Terminal: Amidation
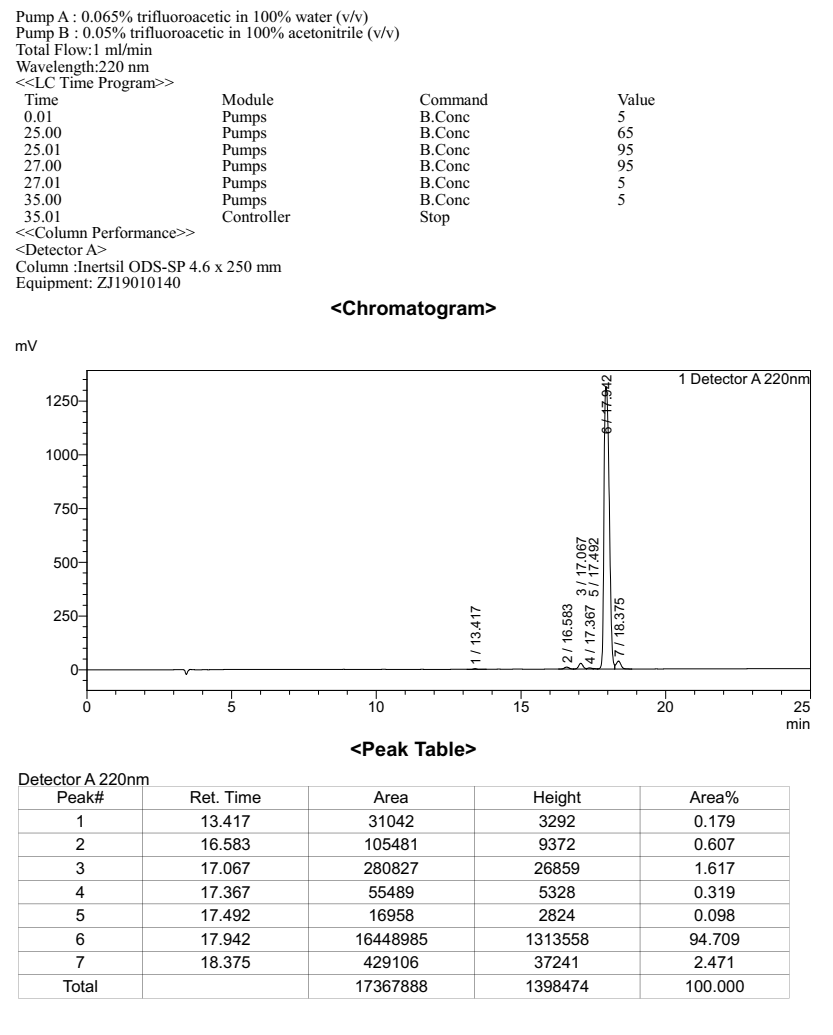
Purity: 94.7%
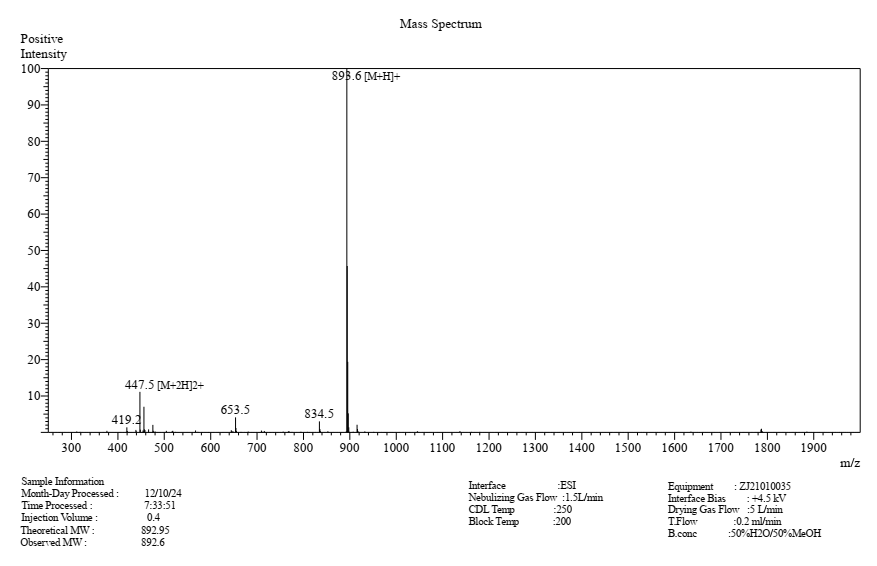
Theoretical MW: 892.95
Observed MW: 892.6
MS result checked by ESI:
HPLC result checked by RP-HPLC, C18 column:
FAQs
-
How many disulfide bonds can you make in peptide synthesis.
GenScript can synthesize peptides with up to 3 disulfide bonds. However, the synthesis of 3-disulfide peptides is extremely difficult and random cyclization is recommended if possible.
-
Can you cyclize my peptide by disulphide bond (cysteine bridge) while my peptide
sequence contains 3 cysteines?
The third singular cysteine will induce ring-opening in a built cysteine bridge and the molecule will be not stable. The production is difficult and needs to be evaluated by PT team.
-
Could you let me know which fluorescent tags are available from GenScript for peptide
modification?
Please kindly refer to the below table
Name Material Material’s CAS Excitation (nM) Emission (nM) Emission color Position MCA 62935-72-2 328 393 Blue N-terminal Abz 150-13-0 320 420 Blue N-terminal AMC 167690-53-1 360 - 380 440 - 460 Blue C-terminal EDANS 50402-56-7 340 490 C-terminal Dansyl Dansyl 605-65-2 342 562 Green N-terminal FITC-Ahx 3326-32-7 494 521 Green N-terminal FAM 5-Carboxyfluorescein (5-FAM) 76823-03-5 495 520 Green N-terminal 5-TMR 5-TAMRA(5-TMR) 91809-66-4 542 568 Orange N-terminal Cy3 Sulfo-Cy3-NHS 1518643-34-9 555 570 Yellow N-terminal Sulfo-Cy3-Mal 1656990-68-9 N-terminal Cy5 Sulfo-Cy5-NHS 2230212-27-6 646 662 Red N-terminal Sulfo-Cy5-Mal 2242791-82-6 N-terminal Cy5.5 Sulfo-Cy5.5-NHS 673 707 Near-infrared N-terminal Sulfo-Cy5.5-Mal N-terminal Cy7 Sulfo-Cy7-NHS 1603861-95-5 750 773 Near-infrared N-terminal Sulfo-Cy7-Mal 2183440-60-8 N-terminal -
Does Genscript sell fluorescent labeled synthetic peptides with quencher?
FRET pairs
Fluorescence resonance energy transfer (FRET) is a mechanism that describes the energy transfer between two fluorophores. Since FRET efficiency is partly based on distance between a donor and acceptor molecule, this technique is commonly used for studying enzyme efficiency, protein-protein interactions, or other molecular dynamics (Fig 1).
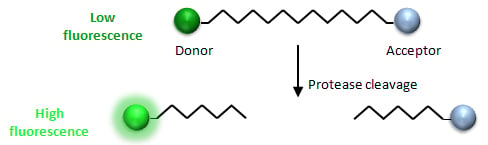
Fig 1. FRET mechanism for protease studies. When the peptide remains intact, the acceptor molecule will quench the donor molecule, and no fluorescence will be detected. If the sequence is cleaved by protease activity, the acceptor will no longer quench the donor, and a fluorescent signal will be detected.
We synthesize the FRET pairs in the below table
Donor Acceptor Name Excitation (nm) Emission (nm) Name Excitation (nm) Emission (nm) Cy2 490 510 Cy3 555 570 FAM 495 520 Cy3 555 570 FAM 495 520 Cy5 646 662 Cy3 555 570 Cy5 646 662 EDANS 335 493 DABCYL 453 - Glu(EDANS)-NH2 335 493 DABCYL 453 - MCA 328 393 DNP 348 - Abz 330 420 DNP 348 - Trp 280 360 DNP 348 - Abz 330 420 Tyr (3-NO2) 360 - -
What is the role of stable isotopic labeling of peptides in NMR spectroscopy?
Nuclear magnetic resonance (NMR) spectroscopy
NMR spectroscopy is a key tool in structural biology research, allowing for the characterization of protein structures, protein dynamics, and protein-protein interactions. It is advantageous over X-ray crystallography, in that the structures of proteins or peptides in liquid can be solved. Additionally, recent advances in NMR spectroscopy have demonstrated its value in drug discovery, due to its ability to help researchers directly identify ligand binding sites.
The technique involves subjecting a sample to a magnetic field, taking advantage of an intrinsic property of certain atomic nuclei, called spin. In response to the magnetic field, nuclear spins flip from low energy spin states to higher energy states. As the spins flip, energy is absorbed by the nucleus. Absorption spectra data can be used to identify atomic nuclei and the distance between nuclei.
Stable isotopic labeling of peptides allows for the incorporation of NMR active nuclei which can help reduce the complexity of spectra and help scientists obtain new correlations between atoms, for more complete structural information.
Selected publications citing stable isotope labeled peptides synthesized by GenScript
 Pustovalova Y. et al. (2012) The C-terminal domain of human Rev1
contains independent
binding sites for DNA
polymerase Η and Rev7 subunit of polymerase Ζ. FEBS Lett. 586: 3051-3056.
Pustovalova Y. et al. (2012) The C-terminal domain of human Rev1
contains independent
binding sites for DNA
polymerase Η and Rev7 subunit of polymerase Ζ. FEBS Lett. 586: 3051-3056. -
How are Click Peptides synthesized?
Our Click Peptides are synthesized using the O-acyl isopeptide method, which was developed by Taniguchi, et al (Figure 1). We use a special synthesis method to incorporate a strategically placed o-acyl bond in place of a naturally occurring n-acyl bond in the native peptide. The resulting peptide, called an o-acyl isopeptide, exhibits enhanced solubility and minimal peptide self-assembly, due to a reduction in intramolecular hydrogen bond interactions between peptide chains.
Adjusting the pH of the Click Peptide solution to ≥7.4 induces a rapid O→N intermolecular acyl migration (conversion of O-acyl bond to N-acyl bond), which rapidly converts the insoluble Click Peptide to its soluble native form. No byproduct is released when the Click Peptide is converted to the native peptide, making it advantageous for in vitro and in vivo biological experimental systems.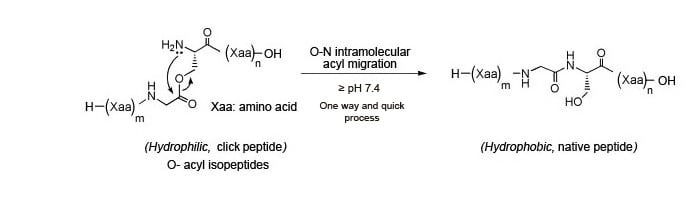
Figure 1: Conversion of Click Peptide to native peptide via pH change
Reference
Atsuhiko Taniguchi, Youhei Sohma, et al. Click peptide: Chemical Biology-oriented analogues of Alzheimer's amyloid β peptide 1–42. J. Peptide Science. Nov 2006; 12(12): 823-828
-
What are the key features of Click Peptides in terms of synthesis and conversion?
Click Peptide synthesis allows control over physicochemical properties (such as water solubility, self-assembly, and folding) and biological activities (like ligand-receptor or enzyme-substrate binding). Additionally, Click Peptides can be easily converted to their native form at pH 7.4 or above through a quick, one-way process with no by-product formation, making them ideal for biological experiments.
-
Can you label my peptide with FITC directly at N-terminus?
If a FITC need to be modified on the N-terminal of the peptide, a linker (Ahx is 6-aminocaproic acid) should be added. If a FITC is modified on the N-terminal amino group directly without a linker, the Edman degradation reaction occurs in the cleavage process.
-
Can you make branched peptides? If yes, how long would each branch be? Can you make
two branches on one original peptide? If yes, how far should two branches separate from each
other?
Yes, we can make branched peptides for you. Usually, the branched peptide should not be too long, 5-10mer would be fine. Our branched peptide is made through Lysine's side chain. We need to evaluate the sequences, if you would like to add two branches onto one original peptide.
-
Are there any requirements for phosphopeptide design?
We recommend that you position the phosphorylated residue no more than 10 residues away from the N-terminus because the coupling efficiency of residues following a phosphorylated residue is significantly reduced
-
Can you make the peptide with aspartic phosphorylation modification?
Unfortunately, we can't make such a modification at present time. We can perform Ser, Thr, and Tyr phosphorylation modification.
-
What is the advantage of capping the N and C termini of the peptide?
Capping will make peptide appear more like native protein. The N terminus can be capped with an acetyl group and C terminus with an amide group.
-
What are the advantages of PEGylation of peptides?
PEGylation adds polyethylene glycol polymer to target molecules through covalent attachments. PEGylation effectively enhances the peptides' therapeutic properties by masking them from the host's immune system, increasing their solubility (for hydrophobic drug GenScript) and bioavailability. It can also prolong the peptides circulatory time within the host through reduced renal clearance.
-
Can a biotin residue be added at the C-terminus of a peptide? Can a spacer be added
at C-terminus to increase the space between the peptide and the C-terminus biotin?
Yes, it can be added in a format of a special amino acid Lys(Biotin), to label it at the side chain of Lys. To add a linker, we could make it as peptide-Ahx-Lys(Biotin).
-
We would like know are you able to do isotopic N and C labeled peptides?
We provide 13C and 15N isotopic labeled peptide. , , , , and are commonly provided isotopic amino acids. Other a.as can be provided according to your demands, but there would be longer the TAT since we need to order raw materials and prices also varies.
-
Can Genscript perform KLH or BSA conjugation on peptide with no cys in sequence?
Yes, we can. There are three methods used to finish protein conjugated peptides. GA method uses an amino group for conjugation, the EDC method uses a carboxyl group for conjugation and the MBS method uses a sulfhydryl group for conjugation.
-
What is the synthesis method for peptoid?
Peptoid Synthesis Method
The synthesis of a peptoid can be achieved through two methods: monomer and submonomer.
Monomer method: This method is very similar to solid phase synthesis of peptide with the exception that all the protected monomers for peptoid synthesis need to be synthesized first, where the synthesis of every Nα-protected monomer is tedious and time-consuming.
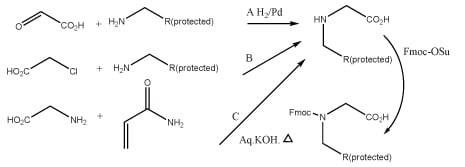
Fig.2 Synthesis of peptoid monomers
Submonomer method: This method eliminates the need of Nα-protected monomers. Each residue is installed in two steps: acylation followed by nucleophilic displacement. The submonomer approach allows the use of any commercially available or synthetically accessible amine that has great potential for combinatorial chemistry. In the acylation step a haloacetic acid, typically bromoacetic acid, is activated by diisopropylcarbodiimide reacting with the amine of the previous residue. In the displacement step (a classical SN2 reaction), an amine displaces the bromide to form an N-substituted glycine residue. This approach has greater advantages over the monomer approach and has been widely applied to peptoid library construction. We currently apply the Submonomer method for peptoid synthesis.
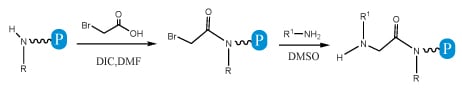
Fig.3 Schematics of peptoid synthesis by submonomer method
-
What is the size of your mini-PEG?
We can provide mini-PEG with two size, which is composed with 2 molecular with 152 Da and 3 molecular with 190 Da respectively. The size we use is depends on the your request. Mini-PEG 2(152 Da) is frequently used.
-
I want to modify a amino acid Histidine (His). The N-terminus needs to be modified by
acetylation (AC group) and C-terminus modified by amidation (CONH2 group). So the chemical
formula is
AC-His-CONH2. Our lab has all the peptide synthesis equipments and In our synthesis, we use
ether to
precipitate peptide after TFA cleavage. I want to ask whether it is possible to precipitate one
single amino
acid in ether.
Generally, Peptides which are less than 6 amino acids and/or very hydrophobic may be difficult to precipitate. An alternative work-up is to evaporate the ether, dissolve the sample in 10% aqueous acetic acid, and extract the organic scavengers and byproducts with chloroform.
-
Why it is difficult to synthesize peptide with FITC without the Ahx linker?
If FITC is directly modified at the n-terminal amino group, the Edman degradation reaction occurs in the cleavage process, as shown in the following figure. Edman degradation is the process of determining the sequence of amino acid residues from the N terminal free of a polypeptide chain. N-terminal amino acid residues were modified by phenyl isothiocyanate, and then the modified residues were cut from the polypeptide chain (the amino acids directly linked to PITC), which were identified by chromatography, and the remaining polypeptide chains (with one less residue) were recycled for the next degradation cycle.
-
Can you synthesize peptides with all D amino acid? If yes, can you provide me a rough
idea of turnaround and cost?
Yes, we can handle such peptides. The synthesis of such peptides is similar to regular peptide synthesis, but due to the low coupling efficiency, the turnaround time will be several days longer than the regular amino acids. The Price is closely related to its specific sequence, we are willing to evaluate it with its specific sequence.
-
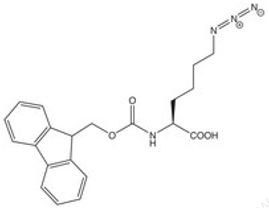
Does your company produce peptides with alkyne or azide amino acids?
At present, we provide peptides service with alkyne and Azide. We can provide {Lys(N3)} and Propargylglycine.
{Lys(N3)}
Propargylglycine
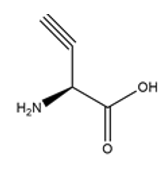
-
How many continuous "R" can we synthesize in a peptide?
We had produced the peptides containing 25 consecutive Arg residues. If you'd like to, we can have a try, but it is hard to ensure success.
-
I also noticed that one of the peptides was pinkish and others yellow.Could you
please let me know the reasons?
Typically, the peptide should be white without any modifications. However, if the peptide undergoes oxidation during transportation, it may appear pink or yellow. If the peptide without modifications is not white upon receipt, please contact us.
-
Can you synthesis peptide with Q at N terminal?
Q at N terminal of peptide is not stable in acidic condition (such as purification and lyophilized) and could undergo cyclization reaction to form pGlu.
To to avoid such as side reaction, N-terminus acetylation is recommended.
Other solutions are 1) Remove "Q" from N-terminus 2) Add another amino acid in front of "Q".
-
How to convert three letter peptide sequence to one letter peptide sequence, or vice
versa?
Please use the following website: http://www.molbiol.ru/eng/scripts/01_17.html
-
Where can we find the full name and abbreviation of peptide modifications?
GenScript can provide multiple modifications, please kindly find the full names and abbreviations of the modifications in the link below.
https://www.genscript.com/peptide_modification.html

-