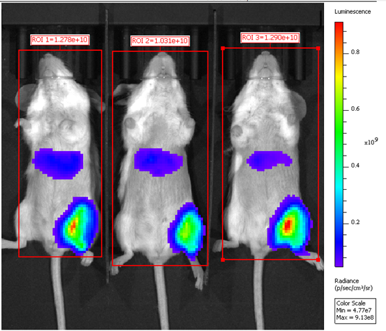
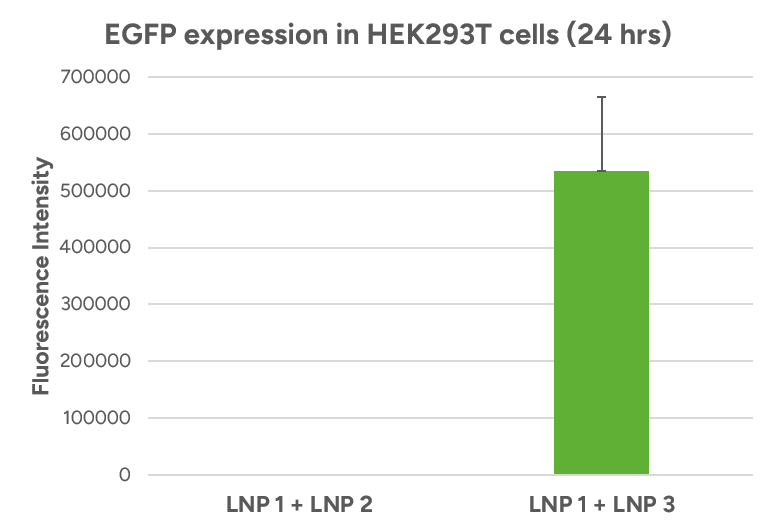
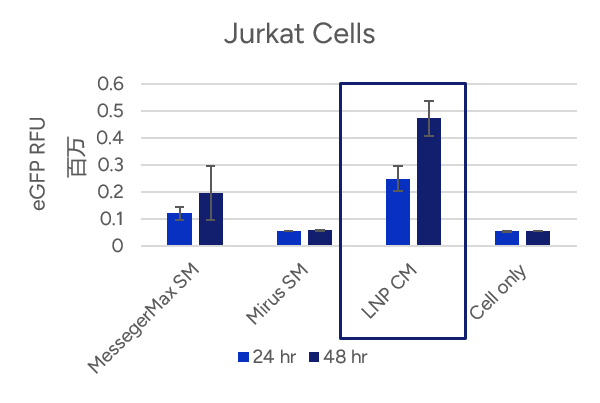
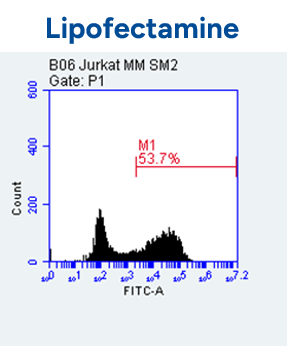
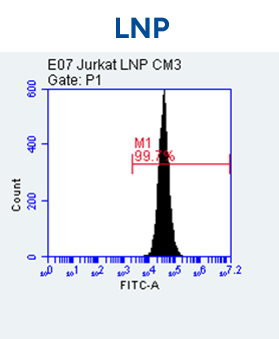
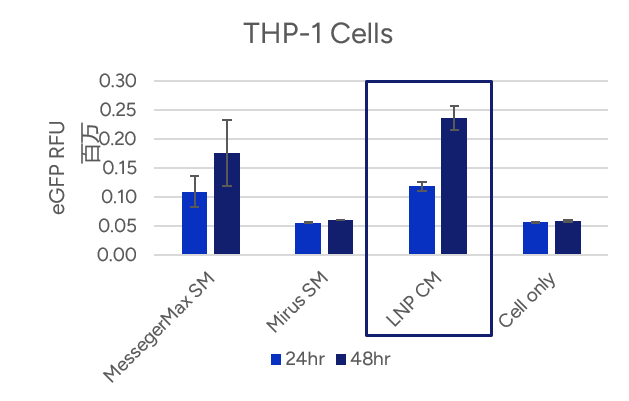
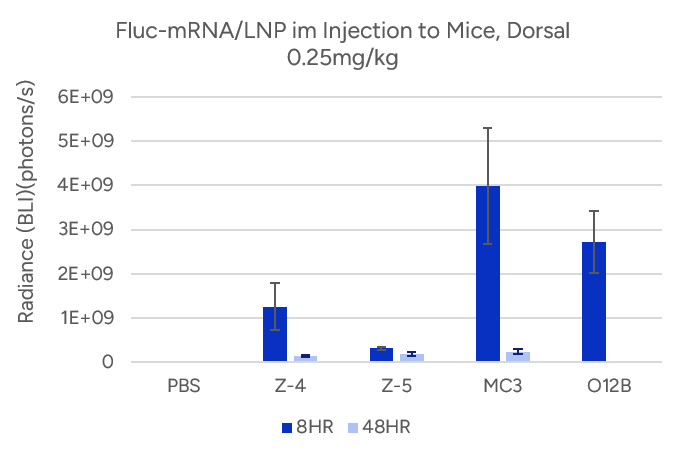
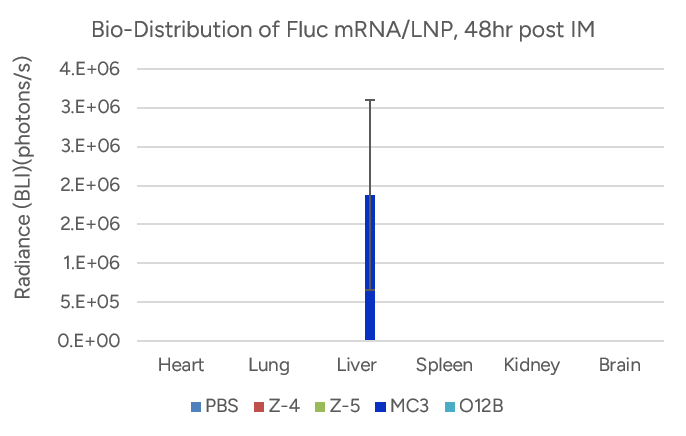
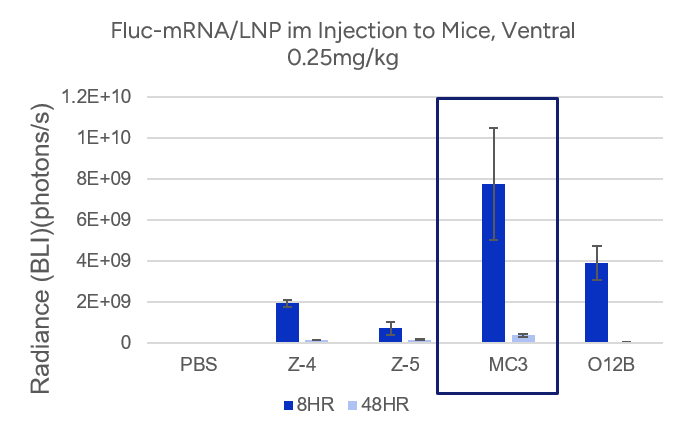
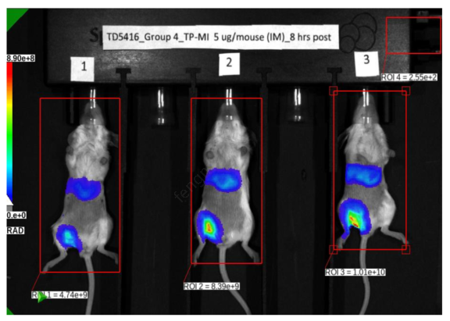
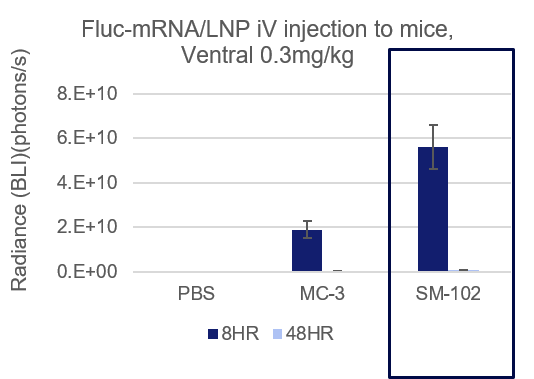
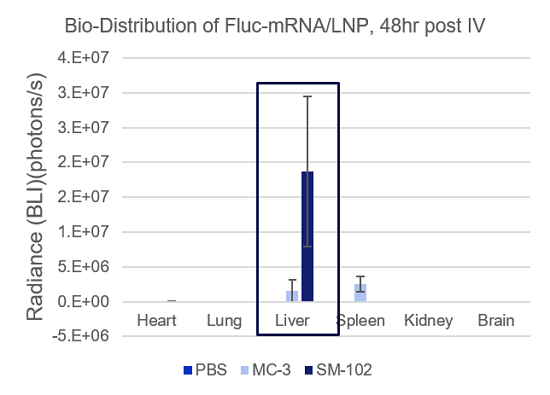
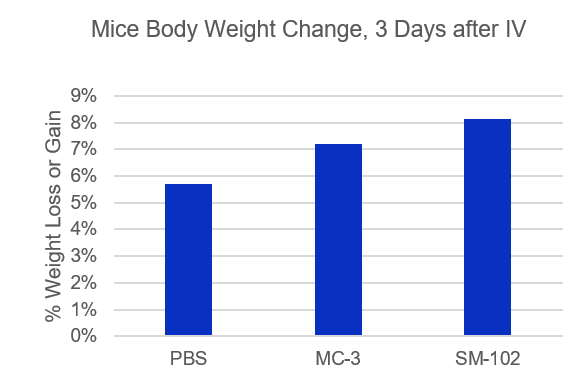
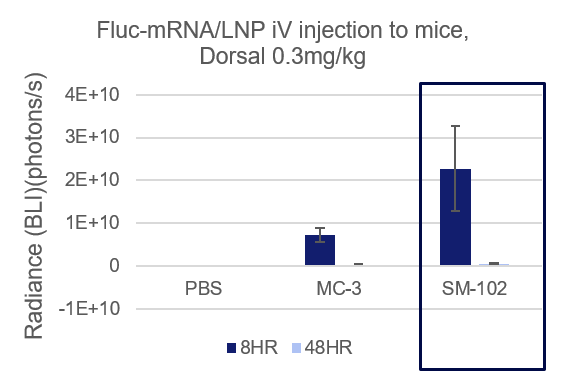
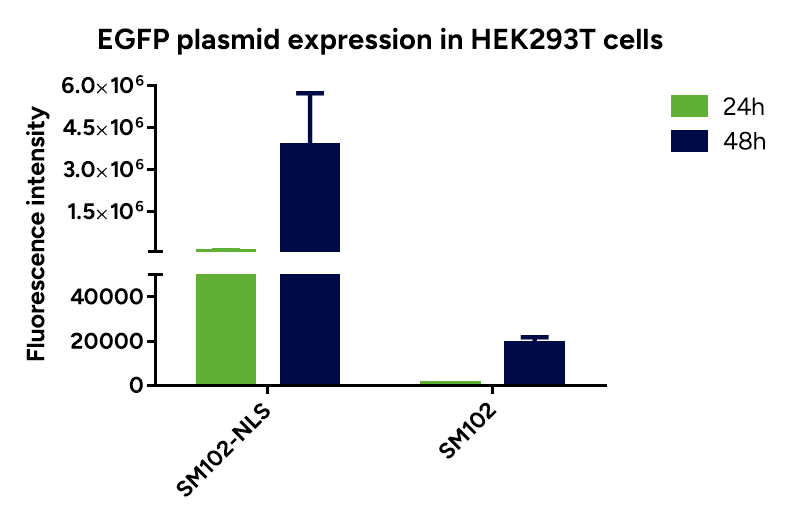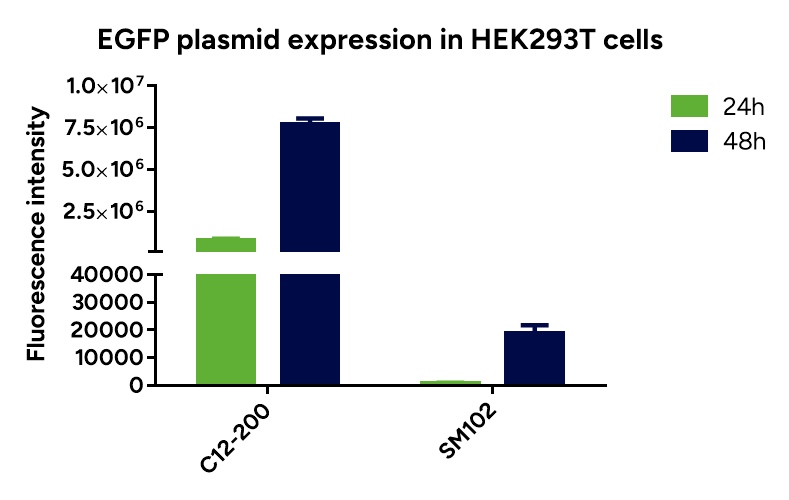- Espcas9 mRNA/sgRNA targeting TARC (GenScript) were formulated in LP01-LNP formulation at 1:1 mass ratio achieved 97% editing efficiency, comparing to 61% by lipofectamine.
- 2.4 ug of RNA/LNP formulation were incubated with HEK293T cells, cell were lysed at day 3 and day 5 for PCR and sanger sequencing.